Acrux Limited is quietly attempting something few small-cap healthcare companies manage successfully. It is using an existing cash-generating business to fund a pivot into a high-value, underserved medical market, without relying on constant capital raises.
The company’s latest update outlines a clear strategic shift toward a Female Testosterone therapy for Hypoactive Sexual Desire Disorder (HSDD), a condition that affects millions of women but remains significantly under-treated.
HSDD is not a niche condition. It is estimated to affect around 10% of adult women, often leading to distress and reduced quality of life.
Yet despite the scale, the US market still lacks an FDA-approved testosterone therapy specifically designed for women.
Around 2 million women in the United States are currently using off-label testosterone treatments, highlighting both demand and the absence of a regulated, purpose-built solution.
Acrux is positioning itself directly into this space.
The company estimates the US menopausal HSDD market alone could be worth approximately US$2.88 billion annually, with broader indications pushing the opportunity closer to US$6.6 billion.
For years, Acrux has operated as a topical generics player, commercialising products using its Metered Dose Transdermal System (MDTS).
That technology, often described as a “patchless patch,” has already been validated in marketed products such as Lenzetto and Evamist.
Now, the company is applying the same platform to a higher-margin therapeutic opportunity.
The Female Testosterone product is already Phase III ready, with FDA-reviewed Phase I and II trials completed, demonstrating efficacy and a favourable safety profile with no material adverse events.
This is not early-stage speculation. It is a late-stage clinical asset with a defined regulatory pathway.
Timing appears to be working in Acrux’s favour.
The FDA clarified the registration pathway for the product in January 2026, reducing uncertainty around approval requirements.
At the same time, regulatory sentiment toward hormone replacement therapies has improved.
The removal of “Black Box” warnings for certain female hormone therapies in late 2025 has helped reshape the risk perception around this category.
CEO and Managing Director John Warmbrunn framed the opportunity in direct terms.
“Female Testosterone is a great opportunity that Acrux is incredibly well positioned to exploit. Our proven capability in registering and commercialising products gives us great confidence that we can gain an early mover advantage.”
Unlike many biotech peers, Acrux is not starting from zero. It already has regulatory, manufacturing, and commercial infrastructure in place through its generics business.
One of the more understated aspects of the story is funding.
Acrux’s existing portfolio is generating increasing licensing income, with quarterly revenues up 305% year-on-year. FY26 licensing income is expected to more than double the prior year.
It means the company can advance a late-stage clinical program without immediate reliance on equity dilution, a common pain point for small-cap biotech investors.
In effect, the generics business is acting as a financial backbone for innovation.
Despite the strategic shift, the market response has been muted.
Acrux shares were trading at $0.011, flat on the day, giving the company a modest market capitalisation of approximately $4.52 million.
Over the past year, the stock has declined 52.17%, reflecting broader small-cap weakness and the market’s cautious stance toward early-stage healthcare plays.
On one hand, the company is targeting a multi-billion-dollar market with a Phase III-ready asset. On the other, it is valued at a fraction of that opportunity.
This disconnect often comes down to execution risk.
Investors might be waiting for clearer milestones, particularly around co-development partnerships expected by mid-2026 and progress toward Phase III trials.
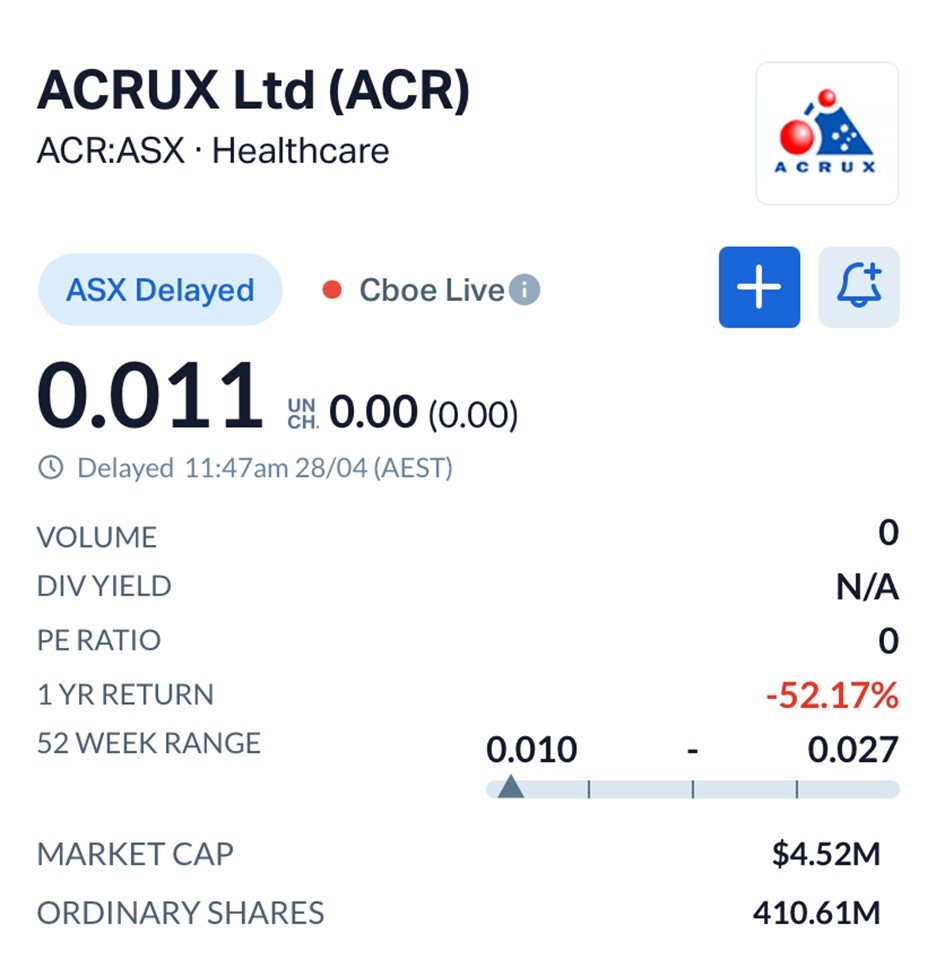
Source: MarketIndex
The broader healthcare landscape is also evolving.
Historically, women’s health has been underfunded relative to other therapeutic areas. That trend is beginning to change, with increasing attention on menopause, hormonal health, and quality-of-life conditions.
Professor Susan R Davis, a global menopause expert, highlights the scientific foundation.
“There is irrefutable evidence that testosterone therapy in doses that result in blood levels within the physiological range for premenopausal women improves sexual desire and reduces personal distress in postmenopausal women with HSDD.”
This growing body of evidence is helping move the conversation from niche treatment to mainstream healthcare priority.
The roadmap is defined but still demanding.
Acrux is targeting a US market launch in FY28 to FY29, with key milestones including:
Each step carries its own risks, but also potential value inflection points.
Acrux is attempting a rare transition, from a steady but low-growth generics business into a high-impact innovator targeting a large, underserved market. The company’s Female Testosterone program offers a compelling mix of late-stage clinical readiness, regulatory clarity, and commercial potential.
Yet the market remains unconvinced, reflected in a subdued share price that suggests investors are waiting for execution, not just promise. If Acrux can convert its Phase III-ready asset into a successful US launch, it may not only unlock significant value but also establish itself as an early mover in a long-overlooked segment of healthcare, where demand is clear, competition is limited, and the opportunity is measured in billions.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles