
Anteris Technologies (ASX: AVR) has entered a defining phase in its push toward the US cardiovascular market after successfully treating the first American patients in its landmark PARADIGM trial for the DurAVR® heart valve.
The procedures were carried out at Montefiore Medical Center in New York by renowned structural heart specialist Dr Azeem Latib, marking the official launch of the company’s pivotal US clinical program.
For Anteris, this is more than another trial update. It is the moment the Brisbane-founded medical technology company steps directly into the world’s largest and most competitive heart valve market, where industry heavyweights such as Edwards Lifesciences and Medtronic currently dominate.
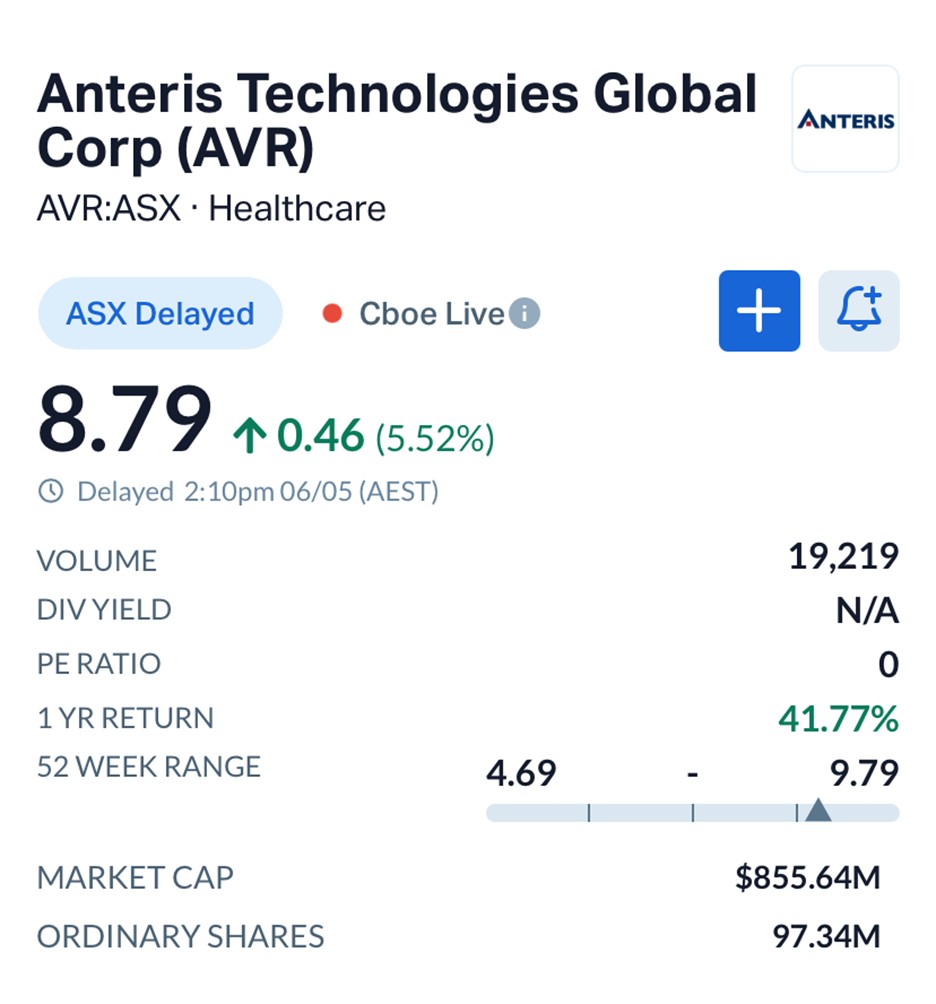
The market responded positively to the milestone, with shares in Anteris Technologies (ASX: AVR) climbing 5.52% to $8.79 by Wednesday afternoon, extending the company’s one-year gain to more than 41%.
Source: MarketIndex
The PARADIGM study is designed as a global randomised controlled trial involving approximately 1,000 patients.
Importantly, the trial is not limited to high-risk or last-resort patients often used in early-stage device testing. Instead, Anteris is enrolling a broad patient population in a direct comparison against commercially available transcatheter aortic valve replacement devices currently used across major hospitals worldwide.
Rather than simply proving the device is safe, Anteris is attempting to demonstrate that DurAVR® can compete with existing market standards on blood flow performance, patient recovery and long-term cardiac function.
The primary endpoint measures a combination of all-cause mortality, stroke and cardiovascular hospitalisation one year after treatment.
The trial will also assess left ventricular recovery and haemodynamic performance, areas where Anteris believes its valve design may offer a clinical advantage.
The company’s core pitch revolves around biomimicry.
Most existing transcatheter heart valves use three separate tissue leaflets. DurAVR® instead uses a single-piece moulded tissue structure designed to replicate the shape and blood flow dynamics of a natural human aortic valve more closely.
The valve also incorporates the company’s ADAPT® anti-calcification tissue technology, which Anteris says has already been used in more than 55,000 patients globally in other medical applications.
That history matters because durability remains one of the biggest long-term challenges in structural heart medicine, particularly as valve procedures increasingly move into younger patient groups.
DurAVR® is delivered through Anteris’ proprietary ComASUR® balloon-expandable delivery system.
Chief Executive Officer Wayne Paterson said the speed of the US rollout reflected strong demand from clinical investigators and hospitals.
“Beginning enrolment within one week of CMS approval demonstrates both the preparedness of our team and the enthusiasm among clinical investigators for DurAVR® THV,” Paterson said.
That rapid activation is notable in a sector where hospital onboarding and procedural approvals can often take months.
The company now plans to expand enrolment across additional global sites as the study progresses.
The transcatheter aortic valve replacement market has become one of the fastest-growing segments in global healthcare over the past decade.
An ageing population, improved minimally invasive procedures and rising rates of cardiovascular disease have driven strong demand for replacement heart valves that avoid open-heart surgery.
According to industry estimates, the global TAVR market is already worth several billion dollars annually and continues to expand as procedures move into lower-risk patient categories.
That opportunity has intensified competition among device makers seeking better durability, improved blood flow and reduced complication rates.
For Anteris, success in the PARADIGM trial could significantly alter the company’s commercial trajectory and strategic value within the global medtech sector.
Historically, strong late-stage cardiovascular trial data has often attracted takeover interest from larger healthcare companies seeking access to differentiated technology platforms.
While the first patient milestone represents an important step forward, the company still faces a long clinical pathway.
The market’s focus will now shift toward patient recruitment speed, procedural outcomes and eventually the one-year data that will determine whether DurAVR® can meet or exceed existing standards of care.
Still, the launch of the US pivotal trial moves Anteris into a different category altogether.
The company is no longer operating as an early-stage concept story. It is now competing directly in the highest-value arena of structural heart medicine, where clinical performance, scalability and physician adoption will determine the next chapter.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles