
The ASX is holding relatively steady on Monday, but one stock drawing attention is 4DMedical, which has officially joined the S&P/ASX 200 following a series of global milestones that highlight its transition into a commercial-stage medical technology company.
The inclusion comes after a week of rapid developments, including a new partnership with pharmaceutical giant GlaxoSmithKline, regulatory clearance in the United Kingdom, and expanded reimbursement pathways in the United States.
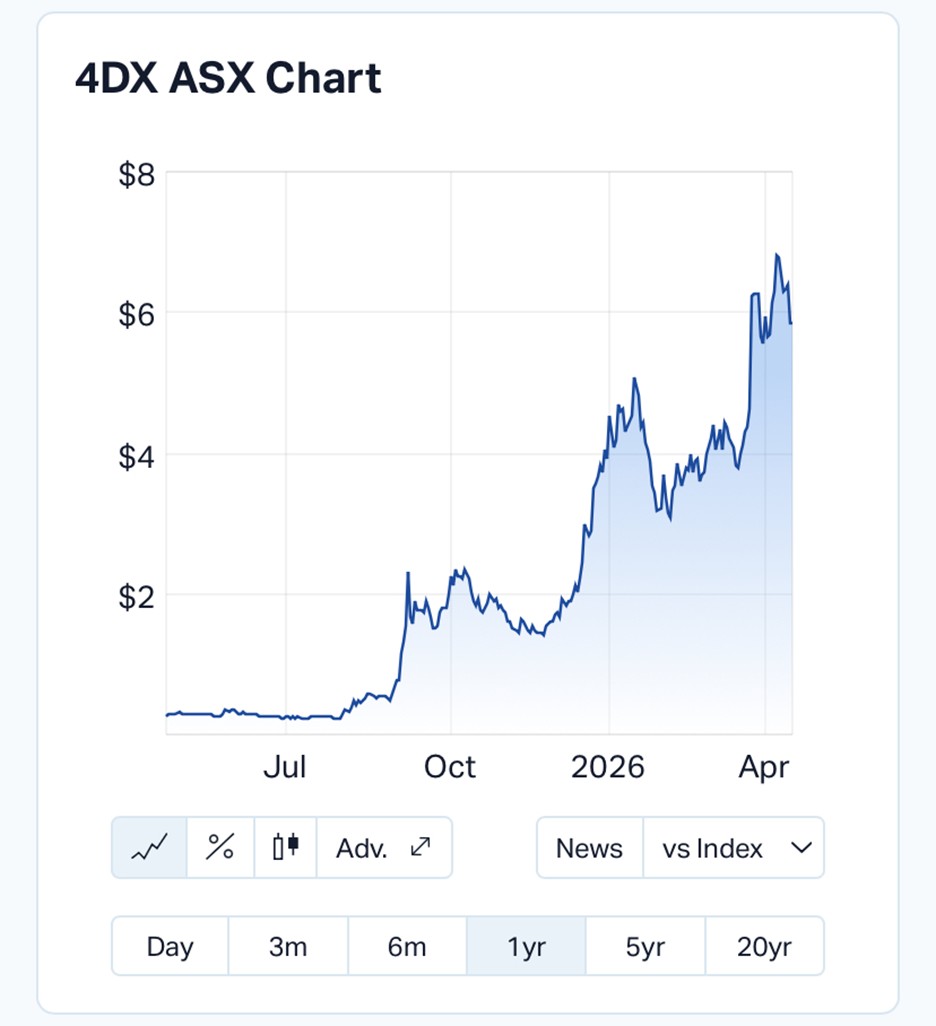
Shares in 4DMedical were last trading at $5.84, holding steady on the day, but the broader trend tells a different story, with the stock up more than 1,900% over the past year.
Source: MarketIndex
The company’s latest update is notable not for a single announcement, but for the timing of several.
At the centre is a one-year contract with GlaxoSmithKline, beginning May 1, which will see 4DMedical provide functional lung imaging biomarkers to support pulmonary drug development and clinical research.
This builds on existing work with AstraZeneca, reinforcing a growing role in pharmaceutical trials where precision and reproducibility are increasingly critical.
In practical terms, this means 4DMedical’s imaging technology is being used not just for diagnosis, but as a measurable endpoint in drug development, an area traditionally dominated by more complex and costly methods.
At the same time, the company has secured UKCA certification for its flagship CT:VQ technology.
This approval allows immediate commercial deployment across both public and private healthcare systems in the United Kingdom, one of the most developed diagnostic imaging markets globally.
Millions of chest CT scans are conducted annually in the UK, spanning respiratory disease, oncology, and emergency care.
By integrating its software into existing CT workflows, 4DMedical is positioning itself as a scalable alternative to traditional nuclear medicine techniques, which often involve higher costs and logistical constraints.
In the United States, the establishment of a new reimbursement code by the Centers for Medicare & Medicaid Services marks another step forward.
The HCPCS code G0680 provides a payment of US$15.50 per study for AI-enabled coronary artery calcium analysis.
While modest on a per-scan basis, the significance lies in creating a defined payment pathway.
Historically, one of the biggest challenges for new medical technologies has been reimbursement uncertainty.
By securing a dedicated code, 4DMedical removes a key hurdle for hospital adoption, making it easier for clinicians to integrate the technology into routine practice.
Further strengthening its global reach, the company has also received clearance from Health Canada for its coronary artery calcium analysis solution.
This adds another major healthcare market to its regulatory portfolio, which already includes approvals across the United States, Europe, the United Kingdom, and New Zealand.
Taken together, these approvals position the company across nearly all major Western healthcare systems.
Despite the scale of the announcements, the stock was relatively unchanged on the day, closing flat at $5.84 after opening at $6.20.
Short-term movements aside, the longer-term trajectory remains strong.
The company has delivered a 41.75% gain over the past month and a 48.6% rise year-to-date, significantly outperforming both its sector and the broader ASX 200.
Its inclusion in the ASX 200 is expected to further increase institutional exposure, as index-tracking funds and large asset managers adjust their holdings.
The broader context is equally important.
Medical imaging has long relied on established methods such as nuclear scans for functional lung assessment.
4DMedical’s approach uses standard CT scans combined with advanced software to generate detailed functional insights, potentially reducing cost, complexity, and patient burden.
This aligns with a wider trend in healthcare, where artificial intelligence is being used to extract more value from existing data rather than relying solely on new hardware.
Industry data suggests that global diagnostic imaging volumes continue to grow steadily, driven by ageing populations and increased screening.
Companies that can integrate into existing workflows while improving outcomes are likely to see stronger adoption.
4DMedical’s latest update reflects a broader evolution within the medical technology sector, where validation is no longer defined by potential alone, but by real-world adoption across multiple fronts.
By securing regulatory approvals, establishing reimbursement pathways, and partnering with global pharmaceutical leaders, the company is moving beyond development into a phase of scalable commercial execution, positioning itself to capture growing demand for AI-driven diagnostic solutions as healthcare systems increasingly prioritise efficiency, precision, and cost effectiveness.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles