Stroke treatment has long been shaped by one brutal reality: every minute matters.
Doctors often use the phrase “time is brain” because delayed treatment can permanently destroy millions of neurons within minutes of a stroke beginning. For emergency departments and ambulance crews, identifying whether a patient is suffering a brain bleed or a clot remains one of the biggest challenges in acute care.
EMVision Medical Devices (ASX: EMV) is now attempting to tackle both problems with the same machine.
The Australian med-tech company announced on Monday it will expand its ongoing emu™ Brain Scanner pivotal trial to include acute ischaemia detection, a move that significantly broadens the device’s potential role in stroke diagnosis and emergency triage.
Shares in EMVision eased 0.66% to $1.895 during afternoon trade, giving the company a market capitalisation of about $176 million.
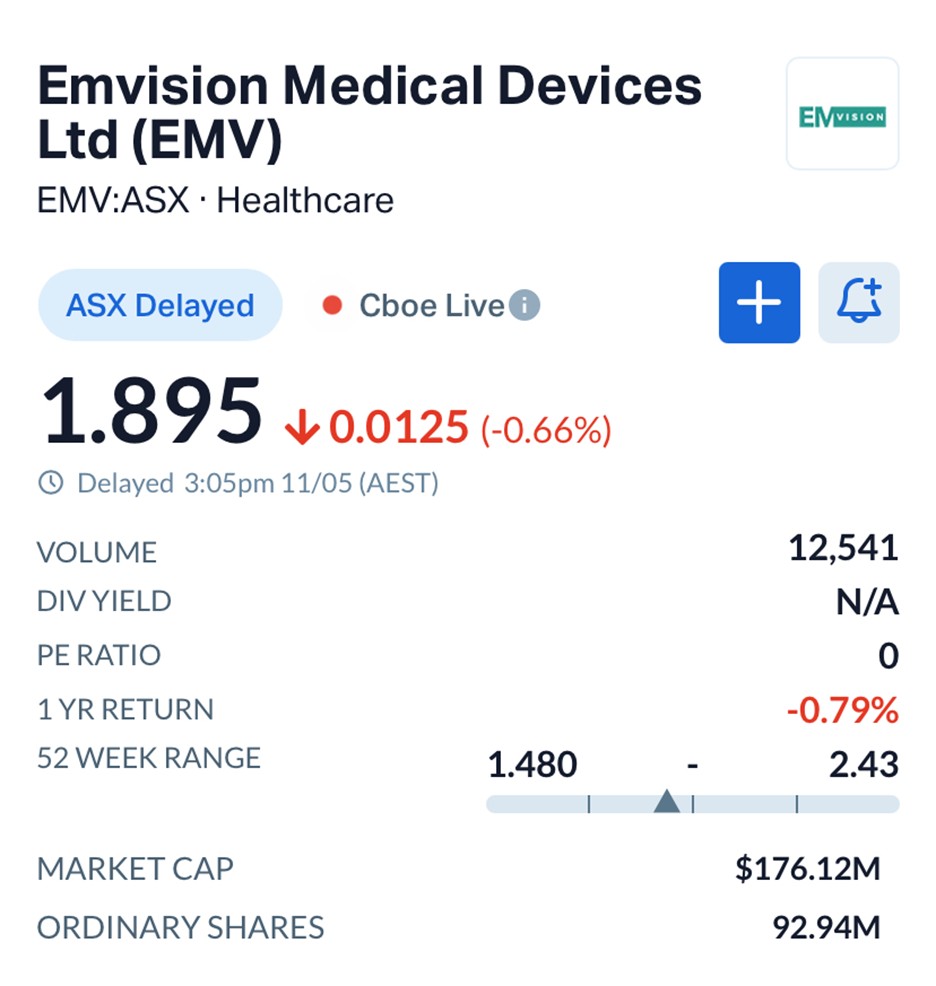
Source: MarketIndex
The announcement marks a major step in the company’s transition from experimental technology developer to potential commercial operator in the global stroke diagnostics market.
Until now, much of EMVision’s focus had centred on detecting haemorrhagic stroke, where bleeding occurs in the brain.
The expanded trial moves deeper into the far larger ischaemic stroke market, which accounts for roughly 80% of all stroke cases globally.
Ischaemic strokes occur when blood flow to the brain is blocked, often by a clot. Treatments such as clot-dissolving drugs and mechanical thrombectomy can be highly effective, but their success depends heavily on how quickly diagnosis occurs.
That timing challenge is precisely where EMVision sees its opportunity.
Its portable emu™ Brain Scanner is designed as a bedside imaging system capable of helping clinicians triage stroke patients earlier, potentially even before they reach advanced imaging suites.
The company says integrating acute ischaemia validation into the existing pivotal trial could save up to two years of development time and avoid the need for a separate, costly standalone study.
Chief executive and co-founder Scott Kirkland described the move as a “step-change” in the commercial and clinical scope of the platform.
“Bringing forward validation of ischaemia detection in the Pivotal Trial is a step-change for our emu™ Brain Scanner,” Kirkland said.
“It expands clinical utility and commercial opportunity from first FDA release, while leveraging the same trial infrastructure for capital and time efficiency.”
One of the more notable developments in the update is how deeply the device appears to be integrating into real-world hospital workflows.
EMVision said the scanner has now been incorporated into “code stroke” pathways at participating hospitals, with no device-related adverse events reported to date.
More than 125 patients have already been recruited across training and primary analysis cohorts.
The pivotal validation trial is currently active across several leading institutions, including Mayo Clinic, Mount Sinai, UCLA, and major Australian stroke centres such as Royal Melbourne Hospital.
Early-stage med-tech companies often struggle to secure access to elite hospital systems, particularly in the United States, where regulatory and operational hurdles can be significant.
The involvement of globally recognised stroke centres adds credibility to both the technology and the trial process.
The bedside scanner itself may only represent the first phase of EMVision’s broader strategy.
The emu™ platform is also acting as the technical foundation for the company’s “First Responder” device, which is being designed specifically for pre-hospital environments such as ambulances and regional emergency services.
In many regional and remote communities, patients can wait hours before accessing advanced brain imaging equipment like CT or MRI scanners.
Portable stroke imaging has increasingly been viewed by clinicians as one of the healthcare sector’s major unmet needs, particularly for rural healthcare systems facing specialist shortages.
EMVision’s Regional Benefit Study in South Australia is being positioned as a possible blueprint for broader deployment in underserved areas globally.
Dr Reade De Leacy, an interventional neuroradiologist at Mount Sinai in New York, recently highlighted the growing need for technologies capable of identifying “true strokes” earlier in the care pathway.
The company believes earlier detection could improve patient outcomes while reducing pressure on overloaded emergency departments.
Globally, stroke care technology has become one of the fastest-growing areas in medical imaging and diagnostics.
Healthcare systems are increasingly investing in portable, AI-assisted diagnostic tools designed to shorten treatment times and improve access outside major metropolitan hospitals.
According to industry estimates, stroke-related healthcare costs exceed US$70 billion annually in the United States alone, while delays in diagnosis remain one of the largest contributors to long-term disability costs.
EMVision is attempting to position itself within that gap.
The company finished the March quarter with $18.4 million in cash reserves and still has access to $6.2 million in remaining non-dilutive funding support after receiving a further $0.4 million grant instalment in May.
Full enrolment for the pivotal trial is targeted for late calendar 2026 or early 2027, with sequential cohort readouts expected thereafter.
For now, the market appears focused on whether EMVision can convert promising clinical momentum into regulatory approval and commercial adoption.
But Monday’s update suggests the company is no longer building a device designed solely to detect brain bleeds.
It is trying to build an all-in-one portable stroke platform for the hyperacute window where minutes can determine whether patients recover, deteriorate, or survive at all.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles