
Neuren Pharmaceuticals (ASX: NEU) is beginning to look less like a traditional biotech company and more like a high-margin pharmaceutical royalty business.
The Australian drug developer delivered another standout quarter on Wednesday, reporting US$101 million in Q1 2026 net sales for DAYBUE, the world’s first approved treatment for Rett syndrome.
The result marked a 20% increase from the same period last year and reinforced growing confidence that DAYBUE’s commercial momentum is not slowing down after its initial launch surge.
Shares in Neuren (ASX: NEU) rose 4.71% to $12.90 by early afternoon trade, giving the company a market value of roughly $1.62 billion.
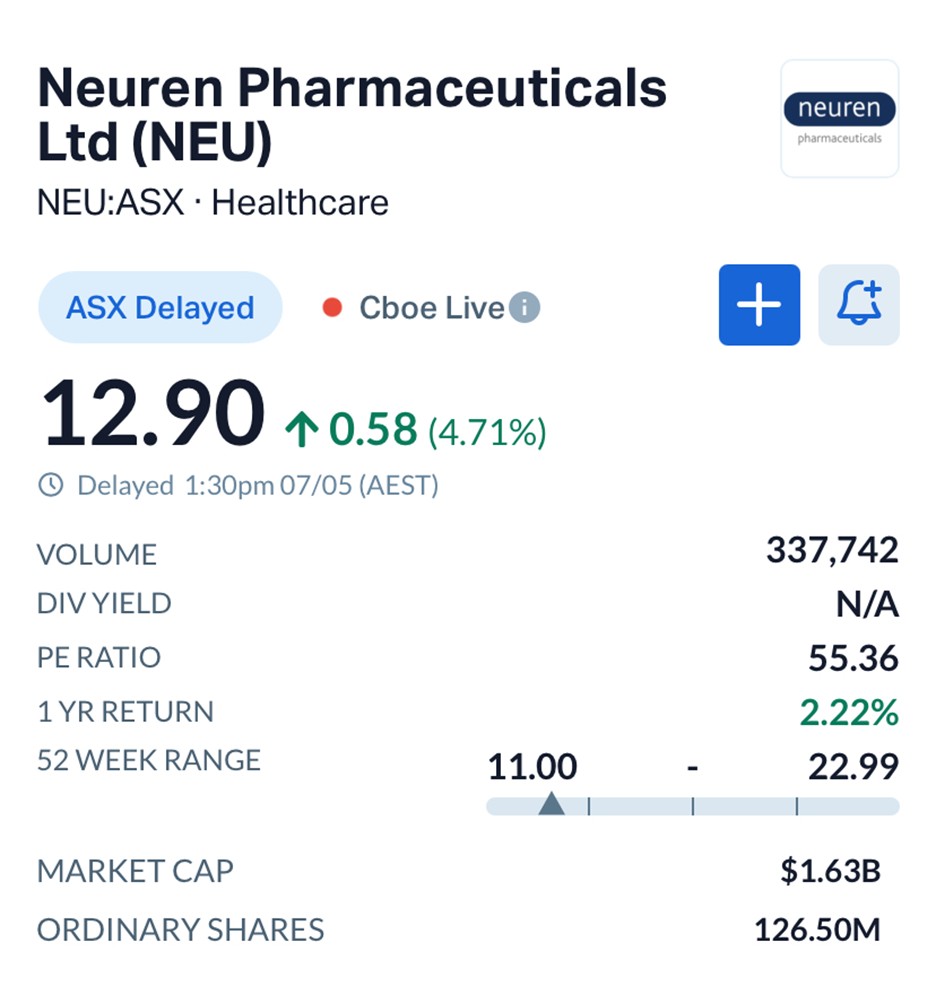
Source: MarketIndex
For a sector where many companies spend years burning cash while chasing clinical milestones, Neuren’s transition into a revenue-generating biotech remains one of the ASX healthcare sector’s rare commercial success stories.
The latest update suggests DAYBUE has now crossed a critical threshold in the biotech lifecycle.
Many orphan disease drugs generate excitement during their first year on the market before sales flatten as patient pools become saturated. Neuren’s latest numbers point to the opposite trend.
Growth is still accelerating.
Neuren earned US$10.4 million in royalty revenue during the quarter, up 23% year-on-year, while partner Acadia Pharmaceuticals reaffirmed full-year DAYBUE sales guidance of US$460 million to US$490 million.
At current exchange rates, that would translate into an estimated A$70 million to A$77 million in royalty income flowing back to Neuren this year alone.
The significance extends beyond headline sales numbers.
DAYBUE’s commercial success is increasingly funding the broader Neuren pipeline internally, reducing the company’s reliance on future capital raisings that often dilute shareholders across the biotech sector.
One of the more closely watched questions surrounding DAYBUE has been whether patient retention would remain stable over time.
Treatments for rare neurological disorders often face practical challenges including tolerability, administration complexity and long-term adherence.
Neuren’s latest update suggests the company may be overcoming some of those hurdles through product innovation.
The company highlighted early momentum from DAYBUE STIX, a new powder-based formulation launched in the United States.
More than 250 prescriptions have already been recorded through the limited rollout.
Importantly, around 30% of those prescriptions came from treatment-naïve patients or patients who had previously discontinued the liquid version.
That detail matters because it suggests the new formulation is expanding the overall treatment pool rather than simply shifting existing users between formats.
Neuren CEO Jon Pilcher said significant room for growth still remains across the US market.
“I see significant potential upside remaining in the US, with penetration rates currently approximately 60% in the Centers of Excellence and only approximately 28% in the broader community,” Pilcher said.
On the new formulation rollout, Pilcher added: “I am very encouraged by the initial uptake and enthusiasm for DAYBUE STIX. I look forward to seeing the impact of the recent broader US launch.”
While the US remains the commercial engine behind DAYBUE, global expansion is beginning to move into sharper focus.
In Japan, Neuren said the trofinetide clinical trial is progressing ahead of schedule, with topline results now expected between September and November this year.
That timeline was previously expected to extend into late 2026 or early 2027.
Meanwhile, the European regulatory review process is approaching a key decision point, with the company expecting a conclusion in June 2026.
Together, those milestones could significantly broaden the addressable market for DAYBUE over the next 12 to 18 months.
The company also received another important validation during the quarter after a Delphi expert panel recommended DAYBUE as part of the standard of care for Rett syndrome.
That recommendation may appear technical, but it carries meaningful commercial implications.
Once therapies become embedded into formal treatment standards, prescription stability and insurance reimbursement often improve materially.
Neuren’s latest update also highlights a broader shift happening across healthcare markets globally.
After years of speculative biotech funding during the low-interest-rate era, investors have increasingly shifted toward companies capable of generating real commercial returns.
The sector’s focus has moved from “potential” toward execution, cash generation and scalability.
Neuren now appears firmly inside that smaller group.
The company is using existing DAYBUE royalties to support development of NNZ-2591, its Phase 3 program targeting Phelan-McDermid syndrome and other neurological disorders.
For many healthcare investors, that transition from “cash burner” to “self-funded innovator” is often where biotechnology companies begin attracting long-term institutional attention.
Rett syndrome remains a rare neurological disorder, affecting mainly young girls and causing severe developmental impairments.
For decades, treatment options focused largely on symptom management rather than addressing the condition directly.
DAYBUE’s commercial performance is therefore being closely watched not only by biotech markets, but also across the broader rare disease pharmaceutical sector.
The latest quarter suggests demand remains strong, patient uptake continues expanding and Neuren’s commercial story may still be in its early innings.
For a company once viewed primarily as a high-risk clinical developer, Neuren Pharmaceuticals (ASX: NEU) is increasingly becoming one of the ASX healthcare sector’s most closely watched revenue growth stories.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles