Pacific Edge spent much of the past year fighting to regain its footing in the United States.
When Medicare coverage for its Cxbladder tests disappeared in April 2025, the New Zealand diagnostics company lost more than reimbursement access. It lost the commercial momentum that had underpinned its long push into the world’s largest healthcare market.
On Friday, the market responded as though that door had reopened.
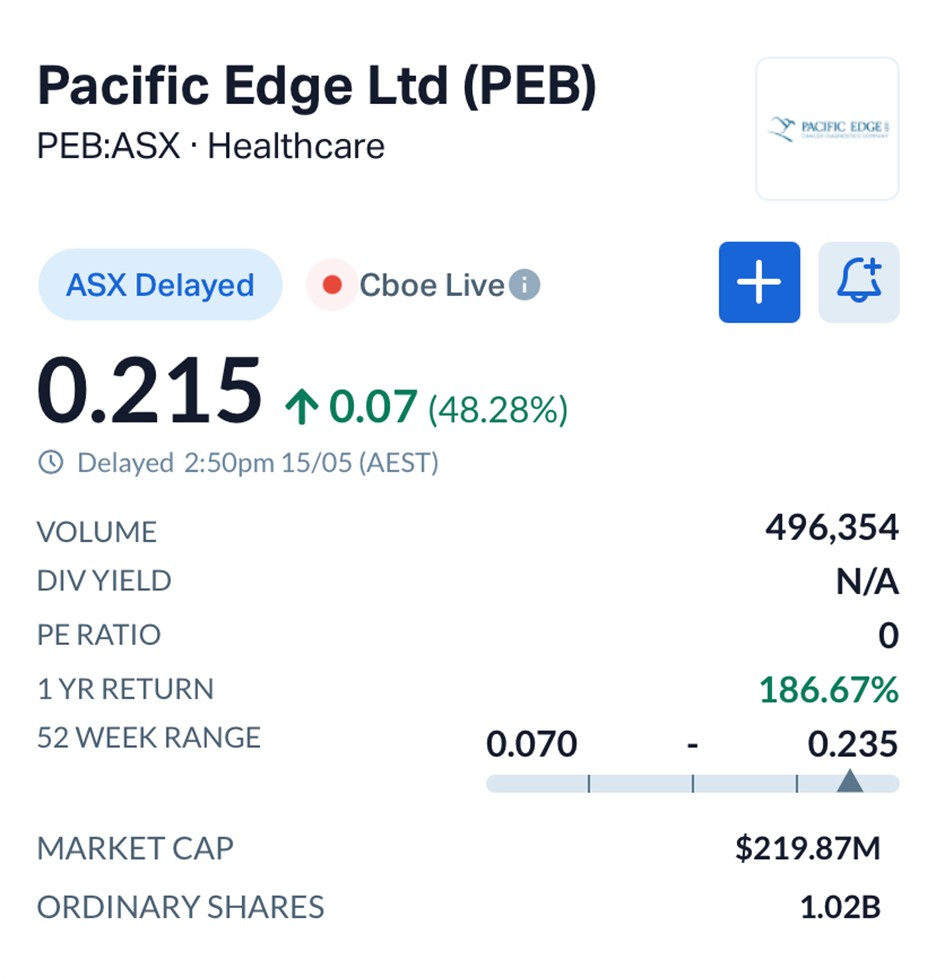
Pacific Edge shares surged more than 48 per cent to 21.5 cents after Novitas, one of the largest Medicare Administrative Contractors in the United States, published a draft Local Coverage Determination proposing coverage for the company’s Cxbladder Triage and Triage Plus tests in patients presenting with microhematuria, or blood in urine, one of the most common warning signs for bladder cancer.
Source: MarketIndex
The move potentially restores a critical revenue stream for Pacific Edge while repositioning the company inside a US diagnostics market increasingly focused on less invasive cancer detection tools.
Volume in Pacific Edge stock climbed sharply through Friday afternoon trade as investors digested the scale of the commercial reset.
The draft Medicare policy, known as DL40378, covers urine-based biomarker tests used during bladder cancer evaluation. Pacific Edge said the proposal positions the company as the only provider currently included for covered urine biomarker testing in the microhematuria setting.
Without Medicare coverage, even clinically supported diagnostics can struggle to gain widespread traction among hospitals and physicians. Coverage decisions from Medicare contractors frequently influence private insurers as well, shaping the broader economics of healthcare adoption across the United States.
Pacific Edge chief executive Dr Peter Meintjes described the draft policy as a major turning point for the company’s long-running US strategy.
“The draft LCD and LCA confirm our position as the first mover and market leader,” Dr Meintjes said.
“Pacific Edge now stands on the cusp of a major commercial inflection point with the only two tests covered for microhematuria evaluation.”
The proposed reimbursement profile also shifts materially higher.
Pacific Edge’s previous Cxbladder Triage test carried a Medicare reimbursement price of about US$760. The newer Triage Plus test sits at US$1,328, reflecting broader clinical applicability and expanded utility across different patient groups.
That pricing dynamic is becoming increasingly important across the diagnostics industry, where companies are under pressure to prove both clinical effectiveness and economic value to strained healthcare systems.
Bladder cancer testing has traditionally relied heavily on cystoscopy procedures, where cameras are inserted into the bladder to investigate abnormalities. Urine-based diagnostics have emerged as an area of growing interest because they may help reduce unnecessary invasive procedures while improving early detection rates.
The broader liquid biopsy and molecular diagnostics market has accelerated rapidly over the past decade as advances in genomics and biomarker analysis reshape cancer screening pathways globally.
Pacific Edge’s turnaround effort has also coincided with fresh capital raising activity.
Earlier this week, the company completed a NZ$25.4 million institutional placement and continues to run a NZ$6 million retail offer for existing shareholders.
That funding now lands against a very different commercial backdrop than it did only weeks ago.
The company had spent much of the past year rebuilding its clinical evidence base and re-engaging with Medicare authorities following the earlier reimbursement setback. Friday’s draft proposal suggests that effort may finally be gaining traction.
For the broader ASX healthcare sector, the announcement also highlights how regulatory and reimbursement milestones can rapidly reshape valuations, particularly for diagnostics companies operating inside the US system.
Pacific Edge shares had traded as low as 7 cents over the past year before Friday’s rally pushed the stock toward fresh 12-month highs. The company’s market capitalisation now sits near A$220 million.
Attention now shifts toward the public consultation period attached to the draft determination and whether final coverage terms remain intact once the process concludes.
For Pacific Edge, the conversation has changed quickly.
Twelve months ago the market was questioning whether the company could rebuild its US business at all. Friday’s move suggests the company may once again be competing for scale inside one of the most commercially valuable diagnostic markets in global healthcare.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles